Allergy Drops
CornerStone Ear, Nose & Throat is proud to be one of the first in the area to offer allergy drops (sublingual immunotherapy). This convenient alternative to allergy shots allows patients to treat allergies by placing drops under their tongue at home instead of getting weekly injections at a doctor’s office.
How Immunotherapy Works
Immunotherapy involves frequently exposing the patient’s immune system to small yet slowly increasing doses of the specific proteins (allergens) that are triggering the allergy symptoms. This process causes gradual desensitization of the immune system to those allergens. Once the body is desensitized, allergy symptoms are typically reduced or resolved.
For many patients, the fear of getting weekly immunotherapy shots overshadows the benefit they would receive. Immunotherapy when medically appropriate is most often declined by patients because of two main factors: fear of needles and/or the inconvenience of actually getting the shots.
For other patients, transportation and time constraints make it impossible to travel to a doctor’s office for weekly allergy shots and then wait for 30 minutes after the injection for safety. The consistent dosing of the allergens is very important, so the inability to go to the doctor’s office consistently is a common reason patients choose not to undergo immunotherapy.
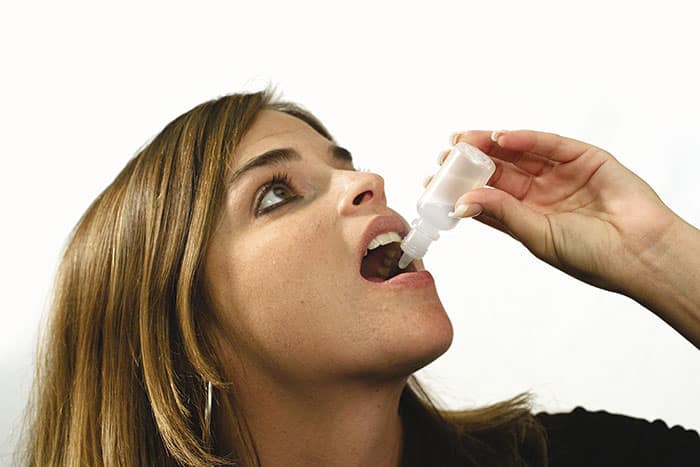
What Are Allergy Drops?
The medical term for allergy drops is sublingual immunotherapy (SLIT). Allergy drops offer all of the same benefits of allergy shots with almost none of the inconvenience or risk factors. Instead of having immunotherapy medication injected through a needle at a doctor’s office, with allergy drops, a patient simply places drops of the same liquid under the tongue daily while at home. SLIT has been endorsed by the World Health Organization as a viable and effective alternative to allergy shots and has been performed internationally for many years, with comparable effectiveness to shots. This allergy treatment is effective for adults and children.
Allergy Drop Facts
- Drops under the tongue replace weekly shots in the office
- A great option for children and busy working adults
- Patients will be seen in the office every 6 to 12 months to monitor their progress
- Antigen extracts for sublingual immunotherapy are the same as those used for allergy shots
- A safe and effective method for treating respiratory allergies caused by airborne allergens including pollen, mold, dust mites, and animal dander






